Let's Explore What We Can Do Together
As a CCRM subsidiary, OmniaBio is building on an established foundation and reputation in manufacturing for cell and gene therapies, expanding on a knowledge base and legacy that has been built over a decade. Together, we are meeting the needs of our CDMO customers. CCRM has collaborated with the following organizations on projects to advance the cell and gene therapy industry.

Nkarta Collaboration Achieves Cost-Effective Manufacturing in Tight Timelines
Nkarta is a clinical-stage cell therapy company focused on engineered...
Case Studies
Immunotherapy
Manufacturing

DiscGenics Collaboration Successfully Develops Allogeneic Therapy to Enable Efficient Manufacturing
DiscGenics is a biopharmaceutical company that develops regenerative cell-based...
Case Studies
iPSC
Process Development

Collaborative Relationship Allows Avectas to Accelerate the Translation of SOLUPORE Into the Clinic
Avectas is an Ireland-based cell engineering technology company developing a...
Case Studies
Immunotherapy
Process Development
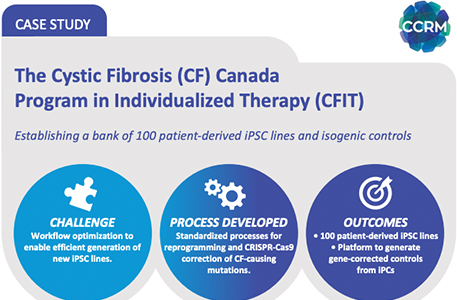
The Cystic Fibrosis (CF) Canada Program in Individualized Therapy (CFIT)
Establishing a bank of 100 patient-derived iPSC lines and isogenic...
Case Studies
iPSC
Process Development

Process Development for Scaled-Up hESC/hiPSC Manufacturing
An optimized 14-day manufacturing workflow developed in < 12 months...
Case Studies
iPSC
Process Development
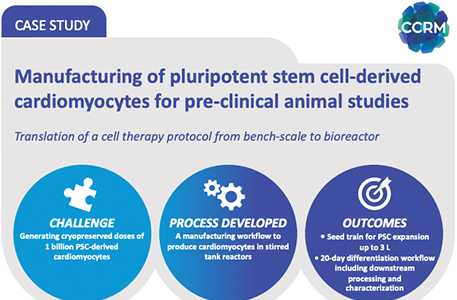
Manufacturing of Pluripotent Stem Cell-Derived Cardiomyocytes for Pre-Clinical Animal Studies
Translation of a cell therapy protocol from bench-scale to bioreactor...
Case Studies
iPSC
Manufacturing
